Our Research
The JTSI group focuses on multiple areas of research, though all topics may be placed under the same broad header of Energy Materials.
Batteries
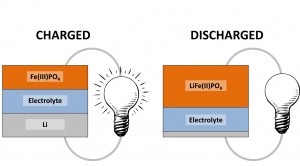 A battery has come to be the name given to an electrochemical power source which is either single use (a “primary battery”) or rechargeable (a “secondary battery”). Classically, a battery is actually a collection of single electrochemical cells (a single cell would simply be called a cell). In these cells there are usually a few different compounds performing different roles. In the simplest case there is an “anode” at the negative terminal, a “cathode” at the positive terminal and an electrolyte separating the two.
A battery has come to be the name given to an electrochemical power source which is either single use (a “primary battery”) or rechargeable (a “secondary battery”). Classically, a battery is actually a collection of single electrochemical cells (a single cell would simply be called a cell). In these cells there are usually a few different compounds performing different roles. In the simplest case there is an “anode” at the negative terminal, a “cathode” at the positive terminal and an electrolyte separating the two.
One of the most prolific secondary batteries is the Li-ion battery. In this case the lithium metal forms the anode and another material which can accept lithium forms the cathode. The cathode material could be FePO4 for example. In this case, when the circuit is complete and the battery under load the lithium metal will oxidise and then move towards the Fe(PO4). This then leads to the formation of LiFePO4 and the simultaneous reduction of the iron.
This leaves one component undescribed; the electrolyte. The choice of material for this component can be very variable but it must be both conductive to electricity and to the ion which is moving across the cell. A number of different cells exist but they all depend on the same principle – the ability of the anode to oxidise and the cathode to reduce leading to a lower energy state (and the commensurate release of energy).
Bio-Energy
Catalysis
Electrolysis
Fuel Cells
Fuel cells are electrochemical devices consisting of an electrolyte and two electrodes capable of converting directly any consumable fuels to electrical energy through the performance of a chemical reaction. The electrochemical process in the fuel cell can be efficient converting nearly 80% of the chemical energy of the fuel into electricity in contrast to the thermal power plants which give efficiency around 40 %. Recently, one technology with advanced potential to achieve the targets, which were included the environment, the energy security, and the radiation/explosion safety, and the advanced technology for electric power generating system and transportation, is strongly considered to the fuel cell technology. Among the various fuel cells, our groups have been focusing on Solid Oxide Fuel Cells and Direct Carbon Fuel Cells to enhance its high conversion efficiency, high power density, flexible choice of fuel such as carbon rich fuels and hydrogen.
Hydrides & Hydrogen Storage
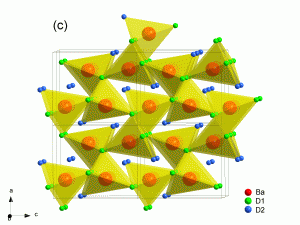 The hydrides of the heavy alkaline earth metals (Ca, Sr, Ba) show high hydrogen mobility in the solid state. The mobile species in these materials is the negatively charged hydride ion (H–) and since the transport properties are predominantly of ionic nature, they would be suitable as electrolyte materials. The hydride ion itself is a strong reducing agent and therefore chemically of great interest. An electrochemical device involving hydride ions could offer new energy efficient ways of synthesising base chemicals, like ammonia or methane from the elements or fully oxidised compounds. Here we try to understand the mechanisms for hydride ion conduction by creating defect chemistry in the parent materials. Various doping strategies are being researched to enhance hydride mobility. New phases are also being synthesised and their structural and electrical properties characterised. Mixed conducting phases would be of particular interest as electrode materials.
The hydrides of the heavy alkaline earth metals (Ca, Sr, Ba) show high hydrogen mobility in the solid state. The mobile species in these materials is the negatively charged hydride ion (H–) and since the transport properties are predominantly of ionic nature, they would be suitable as electrolyte materials. The hydride ion itself is a strong reducing agent and therefore chemically of great interest. An electrochemical device involving hydride ions could offer new energy efficient ways of synthesising base chemicals, like ammonia or methane from the elements or fully oxidised compounds. Here we try to understand the mechanisms for hydride ion conduction by creating defect chemistry in the parent materials. Various doping strategies are being researched to enhance hydride mobility. New phases are also being synthesised and their structural and electrical properties characterised. Mixed conducting phases would be of particular interest as electrode materials.
Nitrogen can also be incorporated into these materials to form a range of nitrided materials, such as nitride hydrides, imides and amides. Exchange of nitrogen and hydrogen within the bulk and with the gas phase seems fast, which are important characteristics in the field of catalysis.
Photocatalysis
Solar energy is inexhaustible in nature and environmental benign. The almost homogeneous distribution of solar incidence worldwide on a daily basis ensures easy accessibility of solar source and wide applicability of solar techniques. Water splitting as well as CO2 reduction into fuels by solar energy is highly promising technology as all the products are now completely renewable and can be readily stored and transported. Unlike photovoltaic which includes expensive fabrication processes, photocatalysis is a very much “cheaper” means of utilising solar energy. It is normally performed in an aqueous solution with a photocatalyst, typically semicondutors in powder form and at ambient conditions. Hydrogen, oxygen, methane and carbon monoxide etc. can be produced gradually with an overall quantum efficiency as high as 56%.
